
Similarly, the “tin cans” that hold soups and other foods are actually made of steel coated with a thin layer of tin. With the increased use of polymeric materials in cars, however, the use of chrome-plated steel has diminished in recent years. 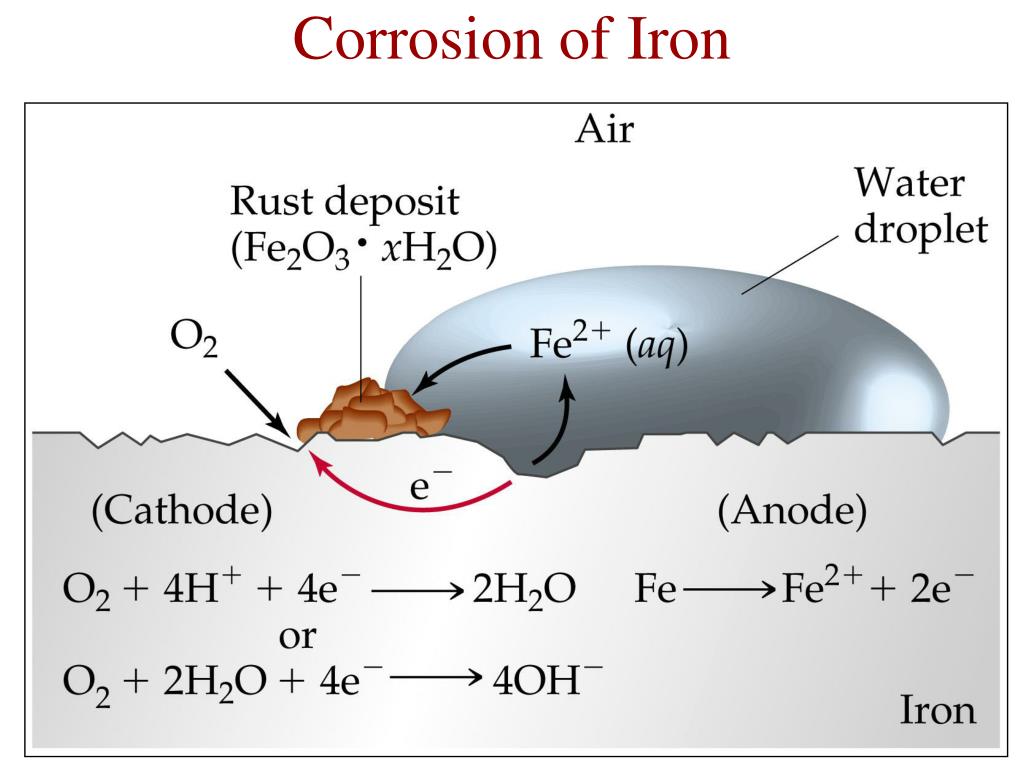
Faucets and some external parts of automobiles, for example, are often coated with a thin layer of chromium using an electrolytic process that will be discussed in Section 19.7 "Electrolysis". One of the most common techniques used to prevent the corrosion of iron is applying a protective coating of another metal that is more difficult to oxidize. The electrochemical interaction between cathodic and anodic sites can cause a large pit to form under a painted surface, eventually resulting in sudden failure with little visible warning that corrosion has occurred. Rust is formed when Fe 2+(aq) diffuses to a location where it can react with atmospheric oxygen, which is often remote from the anode. Holes in a protective coating allow oxygen to be reduced at the surface with the greater exposure to air (the cathode), while metallic iron is oxidized to Fe 2+(aq) at the less exposed site (the anode). Unfortunately, even the best paint is subject to scratching or denting, and the electrochemical nature of the corrosion process means that two scratches relatively remote from each other can operate together as anode and cathode, leading to sudden mechanical failure ( Figure 19.18 "Small Scratches in a Protective Paint Coating Can Lead to the Rapid Corrosion of Iron").įigure 19.18 Small Scratches in a Protective Paint Coating Can Lead to the Rapid Corrosion of Iron (For more information on acid rain, see Chapter 4 "Reactions in Aqueous Solution", Section 4.7 "The Chemistry of Acid Rain".) Automobile manufacturers spend a great deal of time and money developing paints that adhere tightly to the car’s metal surface to prevent oxygenated water, acid, and salt from coming into contact with the underlying metal. Normally, the reaction of atmospheric CO 2 with water to form H + and HCO 3 − provides a low enough pH to enhance the reaction rate, as does acid rain. Under neutral conditions, the driving force is somewhat less but still appreciable ( E = 1.25 V at pH 7.0). The sign and magnitude of E° for the corrosion process ( Equation 19.98) indicate that there is a strong driving force for the oxidation of iron by O 2 under standard conditions (1 M H +). Similarly, a nail immersed in an organic solvent such as kerosene or mineral oil saturated with oxygen will not rust because of the absence of water.Įquation 19.99 4Fe 2+(aq) + O 2(g) + (2 + 4 x)H 2O → 2Fe 2O 3 Because both oxygen and water are required for rust to form, an iron nail immersed in deoxygenated water will not rust-even over a period of several weeks. Instead, the rust continually flakes off to expose a fresh metal surface vulnerable to reaction with oxygen and water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
